21st Austria weekly - Valneva (05/06/2024)
09.06.2024, 1041 Zeichen
Valneva: Valneva, a French/Austrian specialty vaccine company, today announced that the results of two Phase 2 clinical trials of Lyme disease vaccine candidate, VLA15, were published in the peer-reviewed medical journal, The Lancet Infectious Diseases. These trials, as well as a third Phase 2 trial in pediatric participants, supported the design of the current pivotal Phase 3 trial, ‘Vaccine Against Lyme for Outdoor Recreationists’ (VALOR). The Phase 3 clinical trial, VALOR, is currently ongoing to investigate the efficacy, safety and immunogenicity of VLA15 in participants five years of age and older in highly endemic regions in the United States, Canada and Europe. Enrollment of 9,437 participants for the trial was completed in December 2023. Subject to positive data, Pfizer aims to submit a Biologic License Application to the Food and Drug Administration and Marketing Authorization Application to the European Medicines Agency in 2026.
(From the 21st Austria weekly https://www.boerse-social.com/21staustria (05/06/2024)

Wiener Börse Party #1156: Mai-Verfallstag mit zunächst fallendem ATX, Verbund gesucht, am Fenstertag wenige News, aber viel Research
Bildnachweis
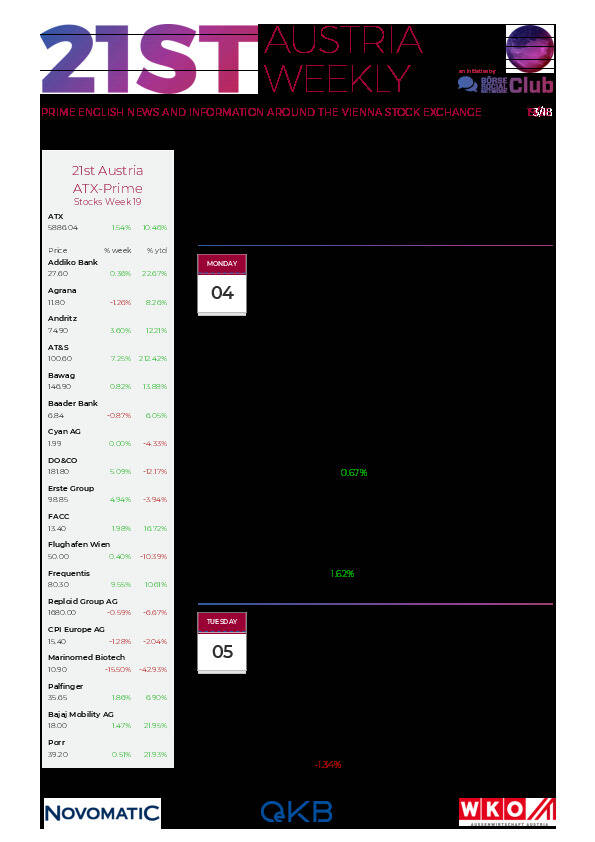
Aktien auf dem Radar:Frequentis, Agrana, Bajaj Mobility AG, EuroTeleSites AG, Amag, UBM, FACC, OMV, Verbund, Porr, Rosgix, EVN, AT&S, Rosenbauer, voestalpine, Wienerberger, Wolford, Wolftank-Adisa, BKS Bank Stamm, Oberbank AG Stamm, CPI Europe AG, Österreichische Post, Semperit, Telekom Austria, RHI Magnesita, DAX, Siemens, HeidelbergCement, MTU Aero Engines, Henkel, Fresenius Medical Care.
Random Partner
UBM
Die UBM fokussiert sich auf Immobilienentwicklung und deckt die gesamte Wertschöpfungskette von Umwidmung und Baugenehmigung über Planung, Marketing und Bauabwicklung bis zum Verkauf ab. Der Fokus liegt dabei auf den Märkten Österreich, Deutschland und Polen sowie auf den Asset-Klassen Wohnen, Hotel und Büro.
>> Besuchen Sie 55 weitere Partner auf boerse-social.com/partner
Useletter
Die Useletter "Morning Xpresso" und "Evening Xtrakt" heben sich deutlich von den gängigen Newslettern ab.
Beispiele ansehen bzw. kostenfrei anmelden. Wichtige Börse-Infos garantiert.
Newsletter abonnieren
Runplugged
Infos über neue Financial Literacy Audio Files für die Runplugged App
(kostenfrei downloaden über http://runplugged.com/spreadit)
per Newsletter erhalten